Xidorin 5 Tablet
By Xidorin
Rx
10 Tablet in a Strip

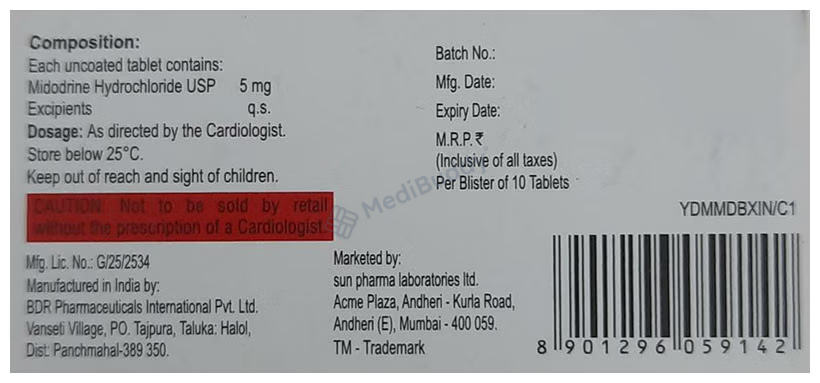
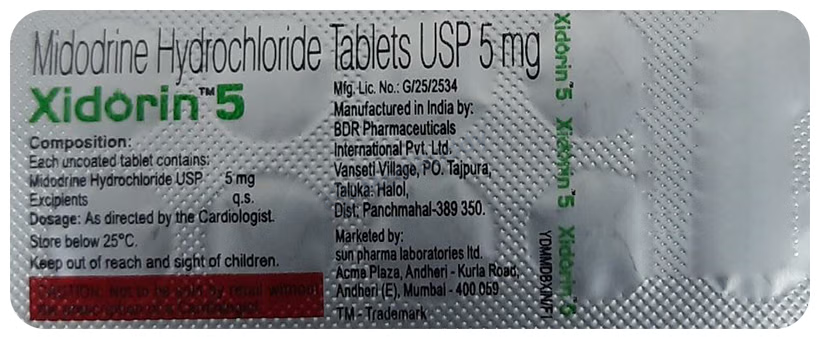
Composition
Midodrine(5mg)

Manufacturer - Sun Pharmaceutical Industries Ltd
Plot no 107/108,Namli block , Ranipool, East Sikkim 737135

Expires on or after
May, 2027

liver
Limited information is available regarding the use of Xidorin 5 Tablet in patients with liver disease. It is important to consult your doctor before taking this medication. Your doctor will assess the benefits and risks associated with using Xidorin 5 Tablet in relation to your liver condition. Please seek professional medical advice before starting treatment with Xidorin 5 Tablet.

kidney
Limited information is available on using Xidorin 5 Tablet in patients with kidney disease. Your doctor will assess benefits and risks before prescribing. Please consult your doctor.

alcohol
Consult your doctor when consuming alcohol with Xidorin 5 Tablet to avoid potential side effects.

driving
Caution is advised when driving while taking Xidorin 5 Tablet. Do not drive if you feel symptoms that may affect your concentration and reactions. Additionally, patients experiencing dizziness or lightheadedness on Midodrine should avoid operating machinery.

pregnancy
Xidorin 5 Tablet should be used with caution during pregnancy as it may not be safe. Animal studies have shown potential harm to the developing baby. Your doctor will carefully assess the benefits and risks before recommending this medication to you. Kindly consult your doctor before using Xidorin 5 Tablet during pregnancy.

breastfeeding
Xidorin 5 Tablet may not be safe for breastfeeding as it could pass into breastmilk and harm the baby. Limited human data is available, so consult your doctor as there is not enough research on its use during breastfeeding.
| Habit Forming | No |
| Chemical Class | Ethanolamine Derivative |
| Therapeutic Class | - |
| Action Class | Alpha-1 adrenoreceptor agonists |
₹739.5
₹870
15% OFF
Inclusive of all taxes
Content verified by

Dr. Archana Prabhakar
MBBS, M.Med (Family Medicine)
Last update on 01-Oct-2024