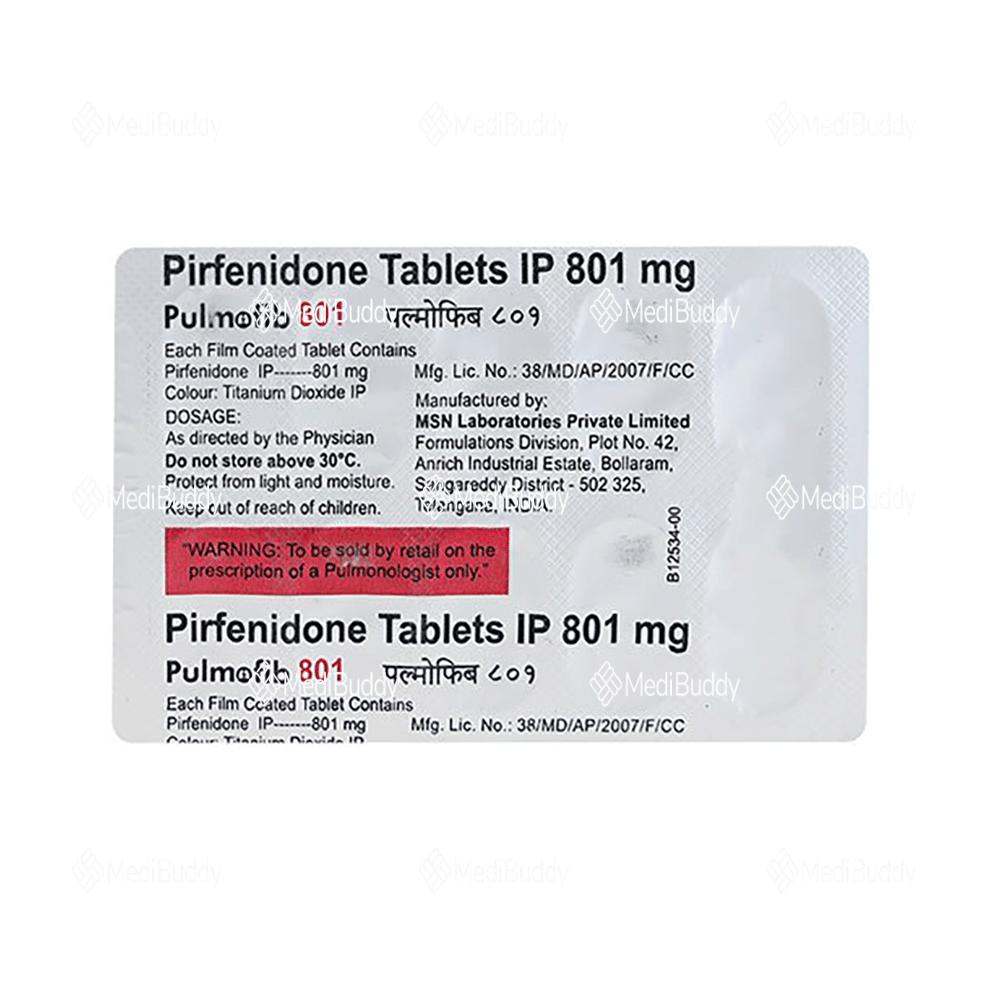
Pulmofib 801mg Tablet
By Pulmofib
Rx
10 Tablet in a Strip

Composition
Pirfenidone(801mg)

Manufacturer - MSN Laboratories
MSN House, Plot No: C-24, Industrial Estate, Sanathnagar, Hyderabad - 18 Telangana, INDIA

Expires on or after
November, 2026

liver
When taking Pulmofib 801mg Tablet, it is important to be cautious if you have liver disease. Patients with liver conditions may require a dose adjustment when using this medication. It is recommended to consult your doctor before starting Pulmofib 801mg Tablet. If you have severe liver disease, it is advised not to use this medication.

kidney
Pulmofib 801mg Tablet requires caution in patients with kidney disease. Dose adjustment may be necessary. Avoid in severe kidney disease and dialysis patients. Consult your doctor.

alcohol
It is uncertain if it is safe to consume alcohol while taking Pulmofib 801mg Tablet. Consult your doctor before drinking alcohol.

driving
For safety while driving, avoid driving if you experience decreased alertness, vision changes, drowsiness, or dizziness while taking Pulmofib 801mg Tablet.

pregnancy
Pulmofib 801mg Tablet may not be safe during pregnancy. Animal studies suggest potential harm to the developing baby. Consult your doctor to evaluate benefits and risks before using it.

breastfeeding
It is better to consult your doctor before using Pulmofib 801mg Tablet.
| Habit Forming | No |
| Chemical Class | Pyridones Derivative |
| Therapeutic Class | - |
| Action Class | Idiopathic pulmonary fibrosis (IPF) |
₹725
Inclusive of all taxes
Content verified by

Dr. Mansi Chaudhari
MBBS - General Medicine
Last update on 01-Oct-2024