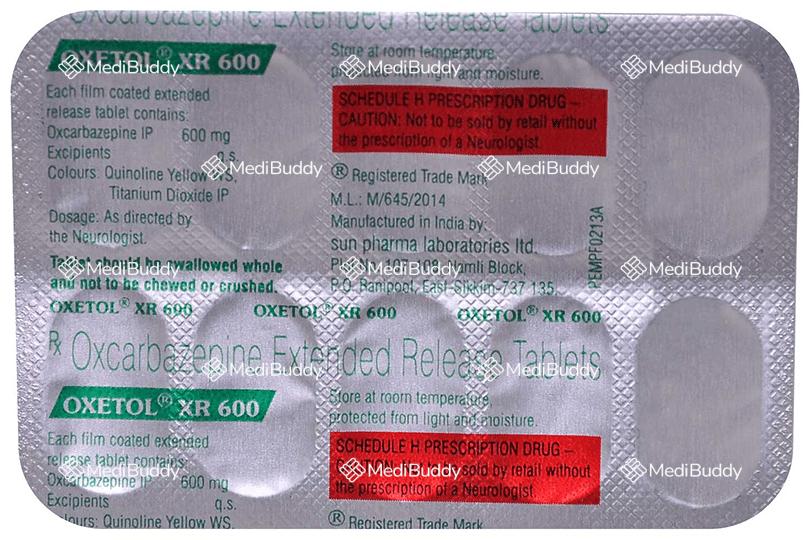
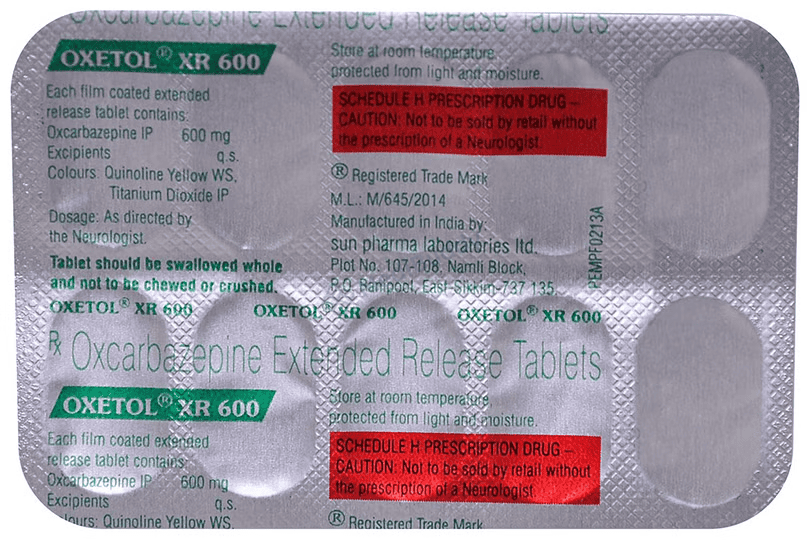
Oxetol XR 600 Tablet
By Oxetol
10 Tablet ER in a Strip

Composition
Oxcarbazepine(600mg)

Manufacturer - Sun Pharmaceutical Industries Ltd
Plot no 107/108,Namli block , Ranipool, East Sikkim 737135

Expires on or after
April, 2027

liver
When taking Oxetol XR 600 Tablet, it is crucial to be cautious if you have severe liver disease. Your doctor may need to adjust the dosage of the medication accordingly. It is important to consult your doctor for individualized guidance. However, for patients with mild to moderate liver disease, dose adjustment is typically not required. Always seek medical advice to determine the most suitable approach for your specific condition.

kidney
Oxetol XR 600 Tablet requires caution in patients with kidney disease. Dose adjustment might be necessary. Consult your doctor.

alcohol
When taking Oxetol XR 600 Tablet, be cautious as it might lead to increased drowsiness when consumed with alcohol.

driving
When taking Oxetol XR 600 Tablet, be cautious when driving as it may cause drowsiness, dizziness, blurred vision, or lack of coordination, especially at the beginning of treatment or when the dose is increased. These effects can impact your ability to drive safely.

pregnancy
Oxetol XR 600 Tablet may not be safe during pregnancy. Animal studies suggest potential harm to the developing baby. Your doctor will assess the benefits and risks before prescribing. Please consult your doctor for guidance.

breastfeeding
Oxetol XR 600 Tablet is likely unsafe while breastfeeding. Limited data indicates it may pass into breastmilk, posing a risk to the baby. Monitor the baby for increased drowsiness and weight gain.
| Habit Forming | No |
| Chemical Class | Iminostilbene Derivative |
| Therapeutic Class | NEURO CNS |
| Action Class | Sodium channel modulators (AED) |
₹258
Inclusive of all taxes
Content verified by

Dr. Mansi Chaudhari
MBBS - General Medicine
Last update on 01-Oct-2024