Loteflam Eye Drop
By Loteflam
Rx
5ml Ophthalmic Solution in a Packet

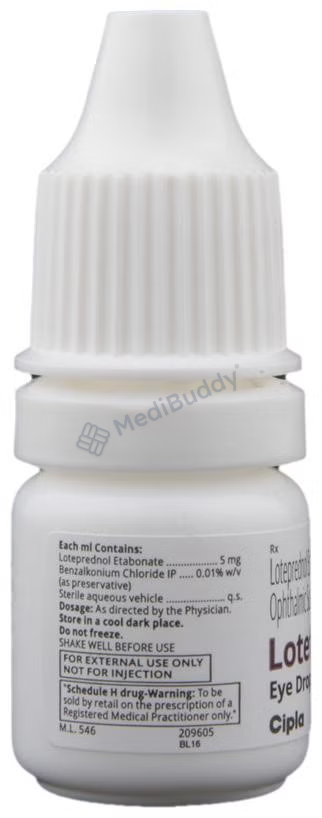
Composition
Loteprednol etabonate(0.5% w/v)

Manufacturer - Cipla Ltd
Cipla House, Peninsula Business Park, Ganpatrao Kadam Marg, Lower Parel, Mumbai-400013

Expires on or after
July, 2026

liver
For individuals concerned about liver issues, it is important to note that no interactions have been found or established with Loteflam Eye Drop. This means that there is no specific caution related to liver issues when using this eye drop. If you have any concerns about how Loteflam Eye Drop may affect your liver or if you have pre-existing liver conditions, it is advisable to consult with your doctor for proper guidance before using this medication.

kidney
No interaction or risks known for kidney issues with Loteflam Eye Drop. If concerned, consult your doctor for guidance.

alcohol
No interaction found between alcohol and Loteflam Eye Drop. The safety advice regarding alcohol consumption with the eye drop is not known. Consult your doctor when cosuming alcohol with Loteflam Eye Drop.

driving
Loteflam Eye Drop may temporarily blur your vision after use. Avoid driving until your vision has cleared.

pregnancy
Loteflam Eye Drop is not recommended during pregnancy as it may not be safe. Animal studies suggest potential harm to the developing baby. Your doctor will assess the risks and benefits before prescribing. Please consult your doctor before using Loteflam Eye Drop during pregnancy.

breastfeeding
Loteflam Eye Drop is considered safe during breastfeeding. Limited data shows no significant risk to the baby. To minimize transfer to breastmilk, apply gentle pressure on the eye and remove excess solution.
| Habit Forming | No |
| Chemical Class | Glucocorticoids |
| Therapeutic Class | - |
| Action Class | Glucocorticoids |
₹163.44
Inclusive of all taxes
Content verified by

Dr. Abdullah Khan
MBBS - General Medicine
Last update on 01-Oct-2024