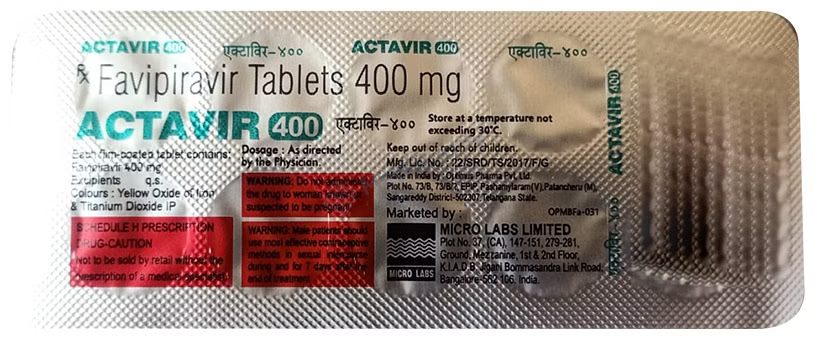
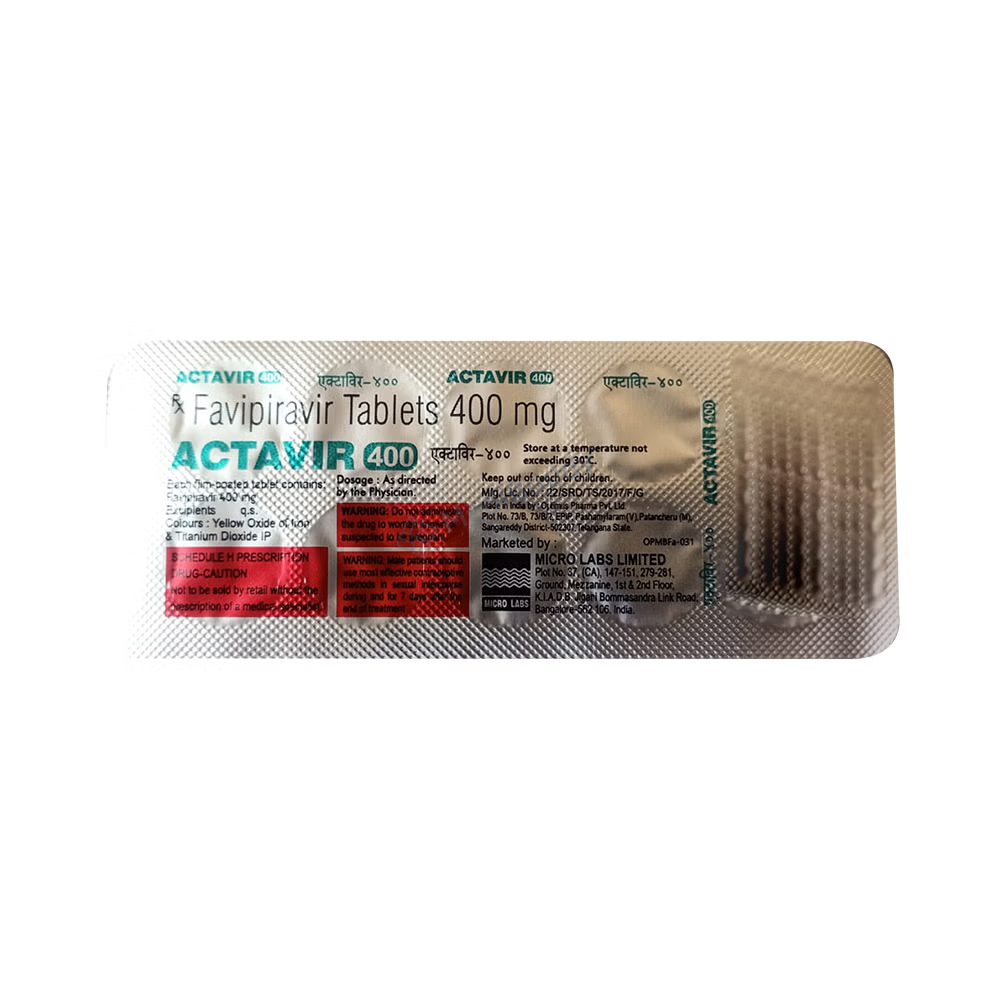
Actavir 400 Tablet
By Actavir
Rx
10 Tablet in a Strip

Composition
Favipiravir(400mg)

Manufacturer - Micro Labs Ltd
27, RACE COURSE ROAD, BANGALORE-560 001, INDIA

Expires on or after
July, 2026

liver
Caution should be exercised when using Actavir 400 Tablet in individuals with severe liver disease. It may be necessary to adjust the dosage of Actavir 400 Tablet for these patients. Please consult your doctor for appropriate guidance regarding the use of this medication.

kidney
Actavir 400 Tablet needs caution in severe kidney disease. Dose adjustment might be required. Consult your doctor.

alcohol
Consult your doctor about consuming alcohol with Actavir 400 Tablet.

driving
When taking Actavir 400 Tablet, be cautious while driving. If you notice any symptoms that make it difficult to focus or react, avoid driving. It's unclear if the tablet affects driving ability.

pregnancy
Actavir 400 Tablet should not be used during pregnancy as it can harm the developing baby, based on studies in pregnant women and animals. Seek advice from your doctor.

breastfeeding
Actavir 400 Tablet is not safe to use while breastfeeding as it may harm the baby. There is a risk of toxicity to the baby when using this medication during breastfeeding.
| Habit Forming | No |
| Chemical Class | Pyrazinecarboxamide derivative |
| Therapeutic Class | ANTI INFECTIVES |
| Action Class | - |
₹890
Inclusive of all taxes
Content verified by

Dr. Gowri Kulkarni
MBBS - General Medicine, DNB - Psychiatry, MRCGP [INT] Family Medicine, BSIC (BACP)
Last update on 18-Nov-2024